Basic characteristics and risk of bias assessment of included studies
As a result, 95 articles were retrieved, and a total of 5 studies (19, 20, 21, 22, 23) with 272 patients were finally included. The article screening process is shown in Figure 1. The five studies were divided into four prospective cohort studies (19, 20, 22, 23) and one retrospective cohort study (21). The details of the five articles are shown in Table 1. A risk of bias analysis was performed on the included studies. Based on the quality analysis, the risk of bias of all studies was acceptable, as shown in Table 2.

Process for identifying eligible research.Our initial electronic search identified 95 studies that met the inclusion criteria and were included in this meta-analysis
survival rate
Three included studies (19, 20, 22) reported the 4-week survival rate of patients and found that TDF did not significantly improve 4-week survival rate compared with the ETV group (RR = 1.17, 95% CI: 0.90) –1.51 , p = 0.24). Five studies (19, 20, 21, 22, 23) provided data on 12-week survival, and combined analysis found no significant difference between the two groups (RR = 1.00, 95% CI: 0.88–1.13, p = 0.94). Two studies comparing 48-week survival in HBV-ACLF (20, 22) showed no significant difference between the two groups (RR = 0.96, 95% CI: 0.58–1.57, p = 0.86) (Fig. 2).

Survival rates of TDF and ETV at 4, 12 and 48 weeks in all included studies
Effect of antiviral treatment on HBV-DNA
Three studies (19,20,21) compared HBV-DNA levels between the two groups at 2 weeks and found no significant difference in HBV-DNA levels between TDF and ETV (SMD = 0.07, 95% CI: -0.55 –0.68, p = 0.83) (Fig. 3A). Three studies (21,22,23) reported unmonitored HBV-DNA rates at 12 weeks and demonstrated that TDF was not effective in improving patients’ HBV-DNA clearance compared with ETV (RR = 1.89, 95% CI: 0.57–6.29, p = 0.30) (Figure 3B).
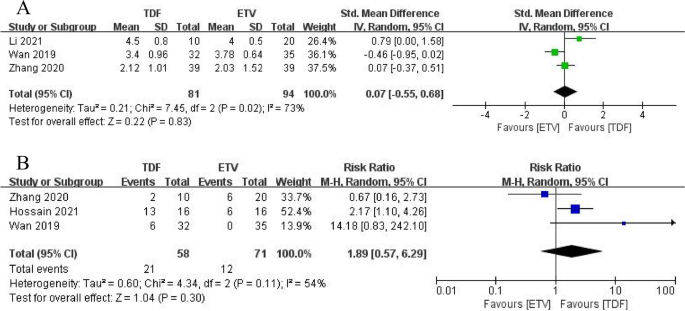
arrive HBV DNA levels decreased in TDF and ETV at 2 weeks. Second HBV DNA clearance rates of TDF and ETV at 12 weeks
Serum surface antigen conversion
In the study by Wan et al. (21) Two and four HBeAg+ patients in the ETV group and TDF group survived for 3 months. Among these patients, 4 patients in the ETV group (0%; 0/2) and the TDF group (100%; 4/4) did not lose HBeAg (p = 0.067). None had HBeAg seroconversion at 3 months. Zhang et al. (22) reported no surface antigen loss in either group at week 48. There was 1 case of HBeAg seroconversion in each group, and the conversion times in the TDF group and ETV group were 12 weeks and 48 weeks respectively.
biochemical reaction
Regarding changes in liver function, three studies (19, 20, 22) examined changes in alanine aminotransferase (ALT) and total bilirubin (TBiL) in the TDF and ETV groups at 4 weeks. Results showed improvement in ALT and TBiL compared with baseline levels, but no significant difference between the two groups (ALT: SMD = 0.65, 95% CI: − 0.04 − 1.34, p= 0.06; TBiL: SMD = − 0.01, 95% CI: − 0.35 − 0.33, p= 0.93). Only one study (22) reported ALT and TBiL at weeks 12 and 48 and found that TDF did not improve ALT and TBiL levels in patients with ACLF compared with ETV (p> 0.05). See Figure 4 for more details.
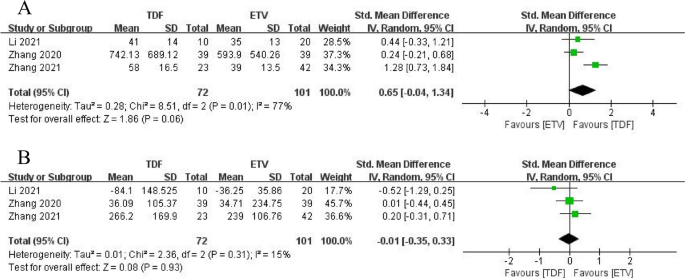
arrive Alanine aminotransferase levels 4 weeks after TDF and ETV treatment. Second Total bilirubin levels 4 weeks after TDF and ETV treatment
CTP score and MELD score
One study (21) and three studies (20,21,22) reported CTP and MELD scores at 4 weeks, respectively. Both CTP and MELD scores belonged to two independent groups (p > 0.05 between baseline, comparable). After 4 weeks of treatment, ETV failed to improve CTP scores or MELD scores, while TDF improved CTP and MELD scores (CTP: SMD = – 0.62, 95% CI: − 1.11 − − 0.13, p= 0.01; MELD: SMD = − 0.72, 95% CI: -1.05 − − 0.39, p< 0.0001). Two studies (21, 23) and three studies (20, 21, 22) respectively mentioned CTP and MELD scores at 12 weeks, and TDF did not improve either score in patients with ACLF compared with ETV (CTP: SMD = -0.75, 95% CI: -2.81–1.30, p= 0.47; MELD: SMD = -1.10, 95% CI: − 2.29 − 0.08, p= 0.07). The forest diagram is shown in Figure 5. Only one study (22) reported MELD scores at 24 weeks, with no significant differences between the two groups (p> 0.05).
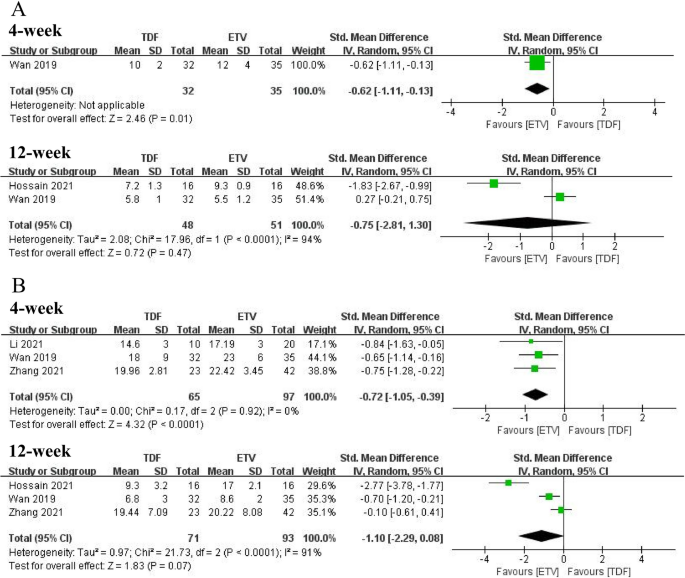
arrive CTP scores for TDF and ETV treatment. Second MELD score for TDF and ETV treatment
Safety of TDF and ETV in HBV-ACLF
Two studies by Zhang et al. and Li et al. (20, 22) provided changes in estimated glomerular filtration rate (eGFR) over 4 weeks but found no significant differences between the two groups in their respective reports. Li et al. (20) focused on eGFR at weeks 4, 12, and 48 and found that eGFR in the TDF and ETV groups declined differently at week 4 compared with baseline, and the decline in the TDF group was greater than that in the ETV group (− 5.83 vs − 4.75 ml/min/1.73m2). However, it is unclear whether the differences in nephrotoxicity are statistically significant. The study by Zhang et al. (19) reported that serum creatine and cystine C were increased in both TDF and ETV groups, but there was no significant difference between them. Furthermore, Wan et al. (21) found no patients with severe lactic acidosis or renal impairment due to ETV or TDF treatment during the 3-month follow-up, whereas Zhang et al. and Li et al. (20, 22) No renal-related adverse events, serious renal adverse events, or proximal tubulopathy events were observed during the 48-week follow-up period, and patients tolerated antiviral treatment well.
Sensitivity analysis and publication bias
We noted considerable heterogeneity in MELD scores and CTP scores at 12 weeks (Yo2 = 94.7% and Yo2 = 91.3%). Sensitivity analysis showed that the study by Hussain et al. is the main source of heterogeneity in the two combined analyses. After excluding this study and combining the analyzes again, no substantial changes were found in the above results, indicating that the results of the meta-analysis are stable.Furthermore, I2 After removing data such as the 12-week survival rate from Wan et al., the value of the 12-week survival forest plot dropped from 55.7% to 28.3%. Begg’s test and Egger’s test found no notable publication bias, indicating that there is no significant publication bias (p= 0.91 (Begg’s test) (Fig. 6).

Begg 12-week survival test. The horizontal line in the funnel plot represents the fixed effects summary estimate, while the diagonal line represents the expected 95% confidence interval given the standard error, assuming no heterogeneity between studies. No publication bias was observed in studies using Egger (p= 0.91) test, indicating no evidence of publication bias

